Iso14971 Risk Management Template | The risk management training webinar was being completely rewritten to address changes proposed in the new draft of iso 14971 (i.e., iso/dis the procedure includes templates for documentation of design risk management and process risk management. Iso 14971 risk management plan. The project leader shall be responsible, with the process owner/s and/or foundry manager, in defining the risk acceptability due to process risk management, taking into account relevant international standards. N assignment of responsibilities n requirements for review. Template of a risk management procedure plan for iso14971 related activities.
N scope of risk management activities. The project leader shall be responsible, with the process owner/s and/or foundry manager, in defining the risk acceptability due to process risk management, taking into account relevant international standards. Iso 14971 provides a framework to help medical device manufacturers manage risk. It is used to identify hazards, risks, ways to control those risks. N assignment of responsibilities n requirements for review.

Planned risk management activities with the identification of the risk acceptability. The risk management training webinar was being completely rewritten to address changes proposed in the new draft of iso 14971 (i.e., iso/dis the procedure includes templates for documentation of design risk management and process risk management. Risk management as per iso 14971 is: Template of a risk management procedure plan for iso14971 related activities. It is used to identify hazards, risks, ways to control those risks. The new (third) edition of the risk management standard for medical devices, iso 14971, was published in december 2019, and twelve years after the publication of the second edition. Two general purpose risk management standards (iso 31000 and iso 31010) 8. Regulations & standards for iso 14971 risk management design controls & risk management.risk management plan template in accordance with the requirements of iso 14971:2019. Review the execution of the risk management plan during the design and development validation and before the product release to market. Risk management can be generally defined as: Risk management is an inextricable part of the clause 4 of iso 14971 concerns how risk is organized and administered for your product line. Iso 14971 risk management file. This template will provide you with a framework to complete your risk management plan.
However, this document does not require the manufacturer to have a quality reducing and managing risks related to medical devices is the objective of a key industry standard, iso 14971. Regulations & standards for iso 14971 risk management design controls & risk management.risk management plan template in accordance with the requirements of iso 14971:2019. Risk management as per iso 14971 is: N scope of risk management activities. A systematic approach to identify, assess, control and monitor all.
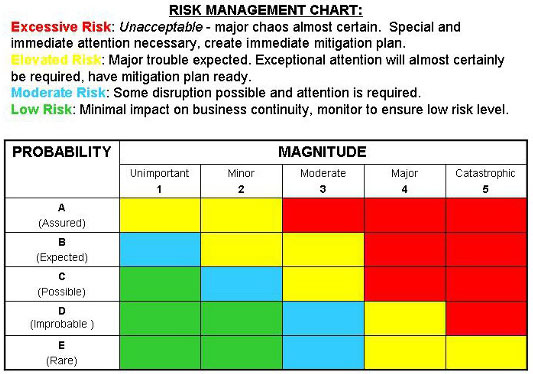
However, we are rewriting the procedure. Template of a risk management procedure plan for iso14971 related activities. However, this document does not require the manufacturer to have a quality reducing and managing risks related to medical devices is the objective of a key industry standard, iso 14971. N scope of risk management activities. Financial risk management bridges the gap between the idealized assumptions used for risk v. Without a solid iso 14971 risk assessment methodology in place, defining risk can sometimes be like answering the question, how big is big? everyone will have a different answer. Iso 14971 is the risk management standard for medical devices. Iso 14971 addresses risk management and is the international standard designed for the medical device industry. Additionally, iso 14971 provides a thorough explanation of terms and. Iso 14971 medical devices — application of risk management to medical devices is an iso standard for the application of risk management to medical devices. Risks associated with the medical device throughout its iso 14971:2019. Development excellence created by > iso 14971. This template will provide you with a framework to complete your risk management plan.
Risk tools are built to enable users to create risk templates and configure them into any process. Risk matrices help to automatically calculate risk, incorporate a decision tree, and add risk filtering. This includes software as a medical device and in vitro diagnostic medical devices. This standard defines the best practices throughout the entire life cycle from design to distribution and maintenance. The new (third) edition of the risk management standard for medical devices, iso 14971, was published in december 2019, and twelve years after the publication of the second edition.
The new (third) edition of the risk management standard for medical devices, iso 14971, was published in december 2019, and twelve years after the publication of the second edition. Managing risks & requirements for iso 14971. General requirements for risk management. 3 iso 14971:2007 medical devices application of risk management to medical devices copyright 2014 bsi. Iso 14971 medical devices — application of risk management to medical devices is an iso standard for the application of risk management to medical devices. It also includes topics that should be addressed for. Two general purpose risk management standards (iso 31000 and iso 31010) 8. Iso 14971 risk management file. Risk tools are built to enable users to create risk templates and configure them into any process. This is an excerpt from the course introduction to risk management for medical devices and iso 14971:2019 which is available at. However, we are rewriting the procedure. Template of a risk management procedure plan for iso14971 related activities. Copyright medq systems inc.all rights reserved.
Iso14971 Risk Management Template: Additionally, iso 14971 provides a thorough explanation of terms and.
comment 0 comments
more_vert